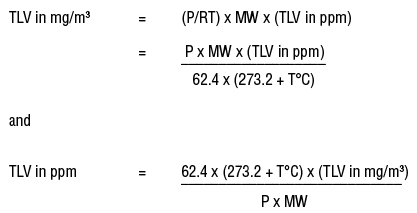
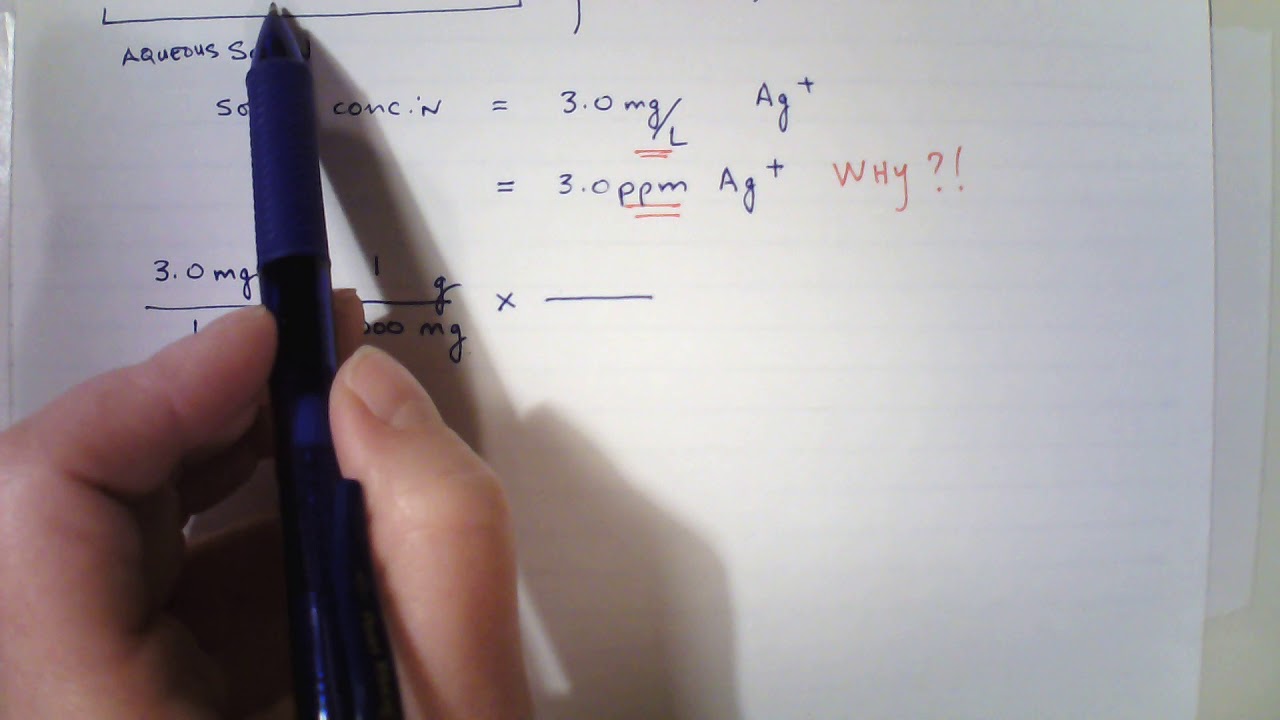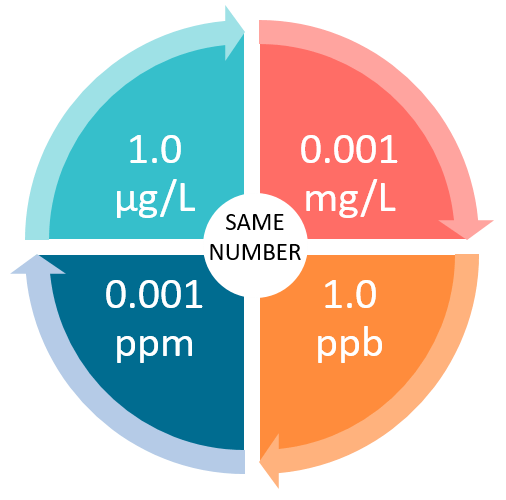Calculate the amount of lime and soda required for the softening of 10^(6) L of a sample of boiler feed water with the following data: CaCO3=1.4^@ Clark, MgCO3=0.56^@ Clark, CaSO4=0.42^@ Clark, MgSO4=0.14^@

White Board Review Practicing with Concentration Expressions Molarity Percent ppm © Mr. D. Scott; CHS. - ppt download
Ozone equipment manufacturer and ozone system integrators Ozone dosage rate into water calculator Ozone Integration Experts

SOLVED: A 190-mg sample of Na2CO3 is dissolved in H2O to give 1.00L of solution. What is the concentration of Na+ in parts per million (ppm)? Note: "ppm" are 106 ×[(mass of

Pounds Formula Prepared By Michigan Department of Environmental Quality Operator Training and Certification Unit. - ppt download