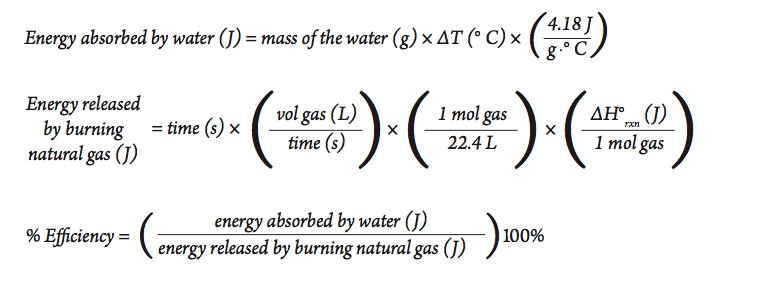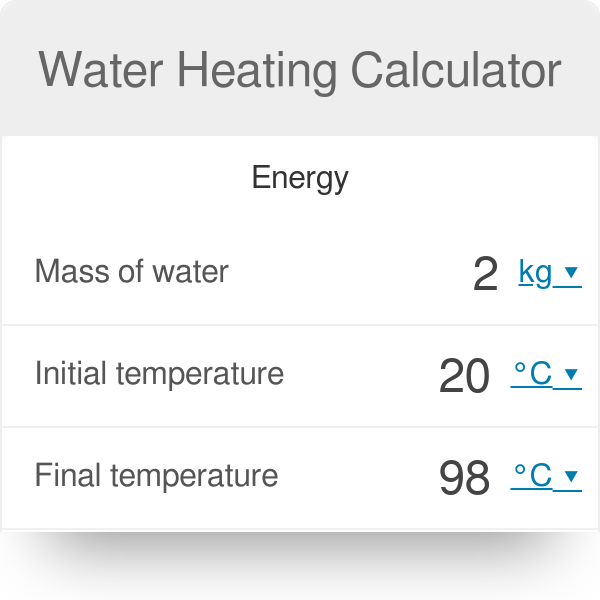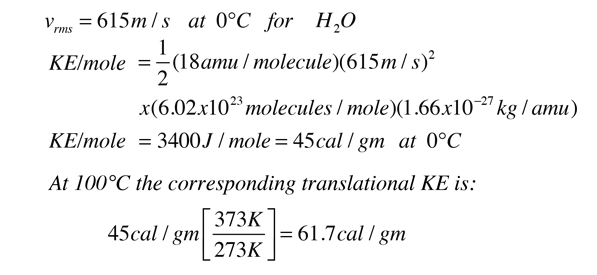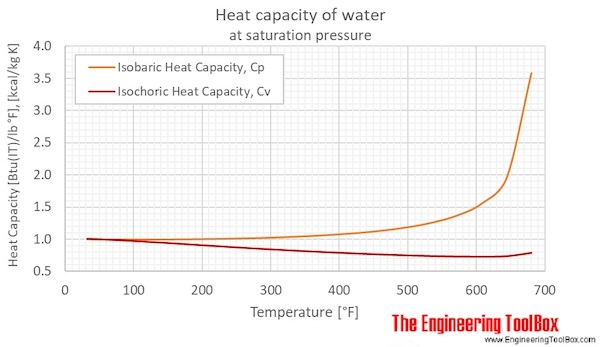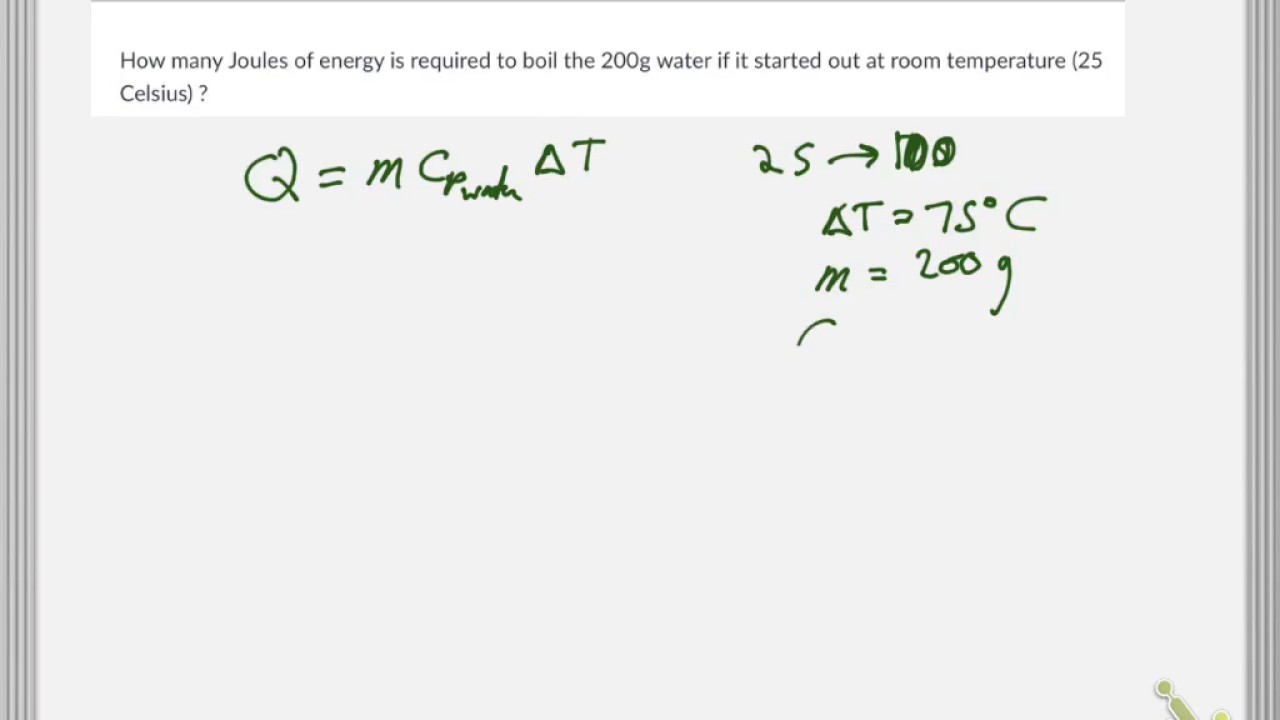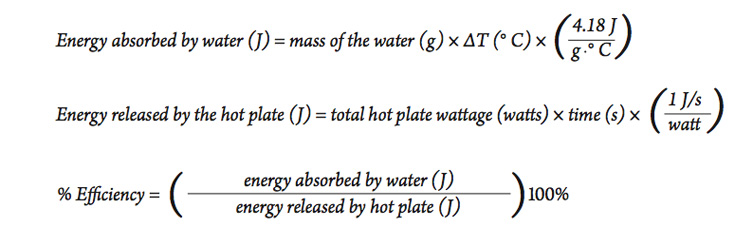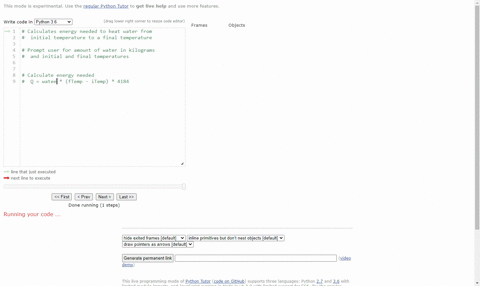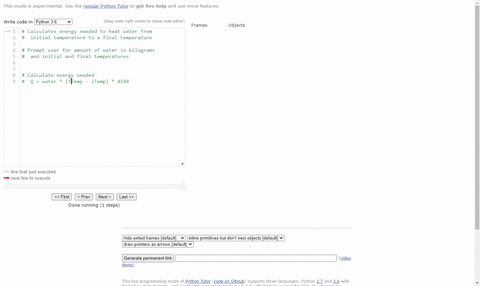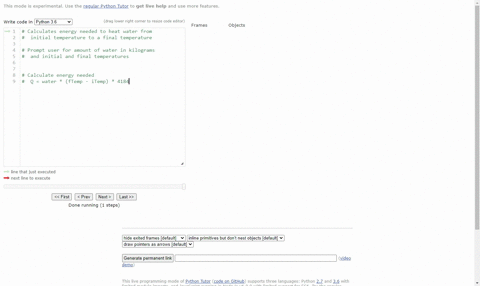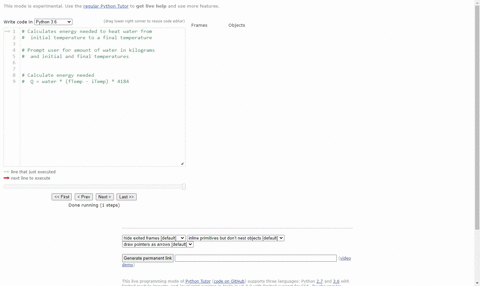
SOLVED:(Science: calculate energy) Write a program that calculates the energy needed to heat water from an initial temperature to a final temperature. Your program should prompt the user to enter the amount
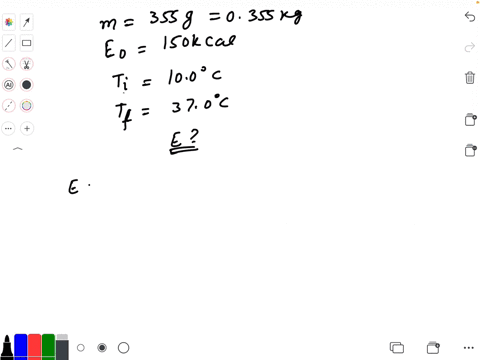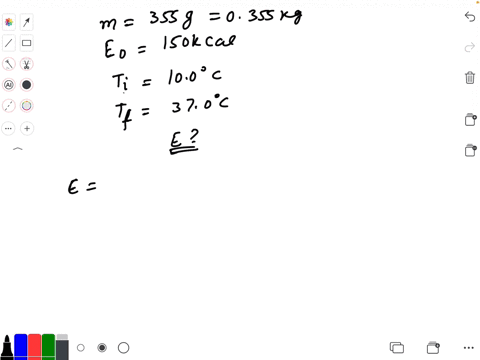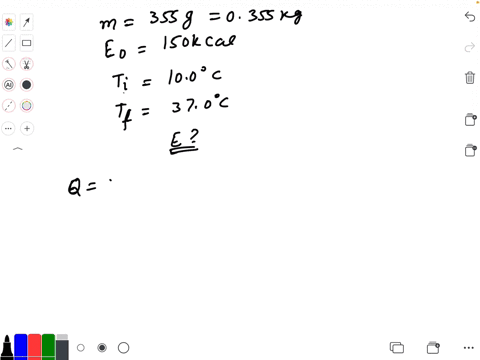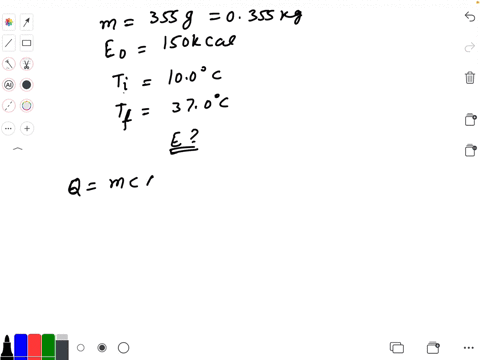
SOLVED: Calculate The Energy Needed To Heat The Cube Of Silver; With A Volume Of Cm? From 14 C To 26 Refer To The Tables Express The Heat In Calories | C&c

Calculate the heat energy required to raise the temperature of 2 kg of water from 10^@C to 50^@C. Specific heat capacity of water is 4200 JKg^(-1) K^(-1)

Question Video: Finding the Amount of Energy Needed to Change the State of Water from Liquid to Gas | Nagwa

Calculating Heat. Specific Heat Amount of heat energy needed to raise the temp of 1 ml of a substance 1°C For water the specific heat is 4.19 J/g °C, - ppt download

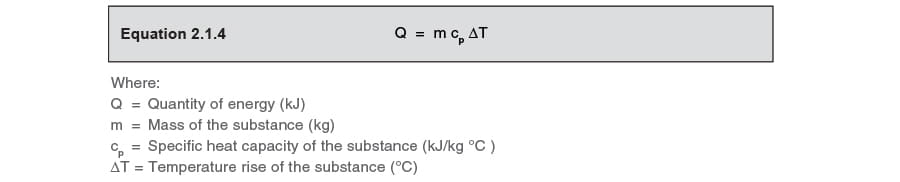
Enthalpy. Specific Heat Capacity Definition: The HEAT ENERGY required to raise the TEMPERATURE of 1kg of substance by 1 o C. e.g. for water C= 4.18kJ. - ppt download