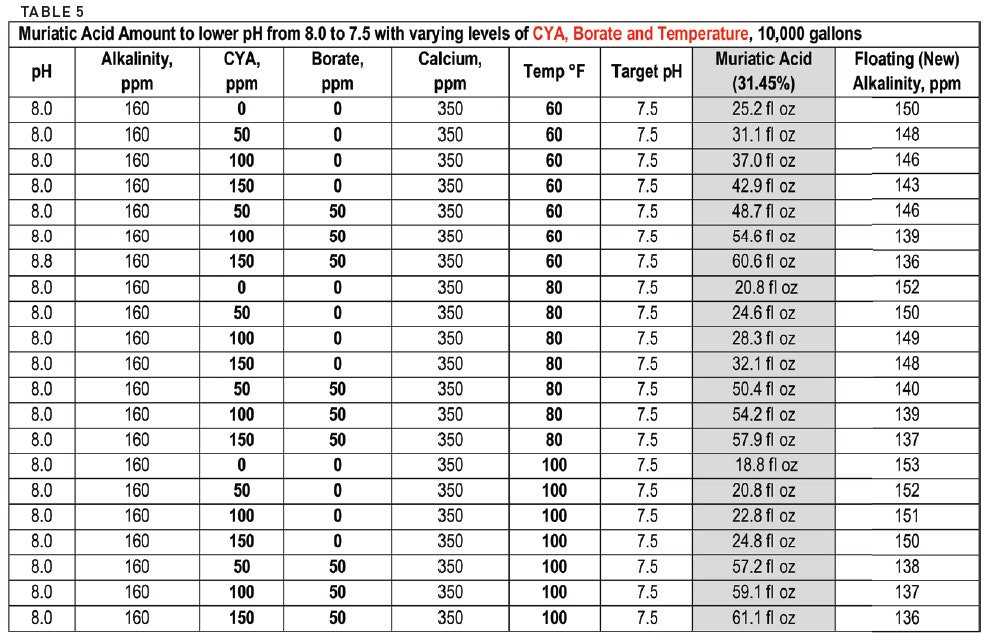
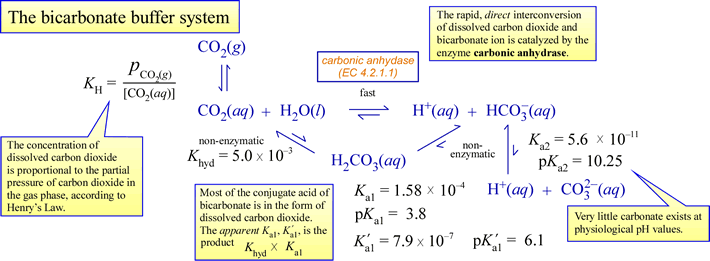
pH calculations and more in fundamentals of pharmaceutics. : Calculate pH and buffer capacity of a carbonic acid/ bicarbonate buffer solution.
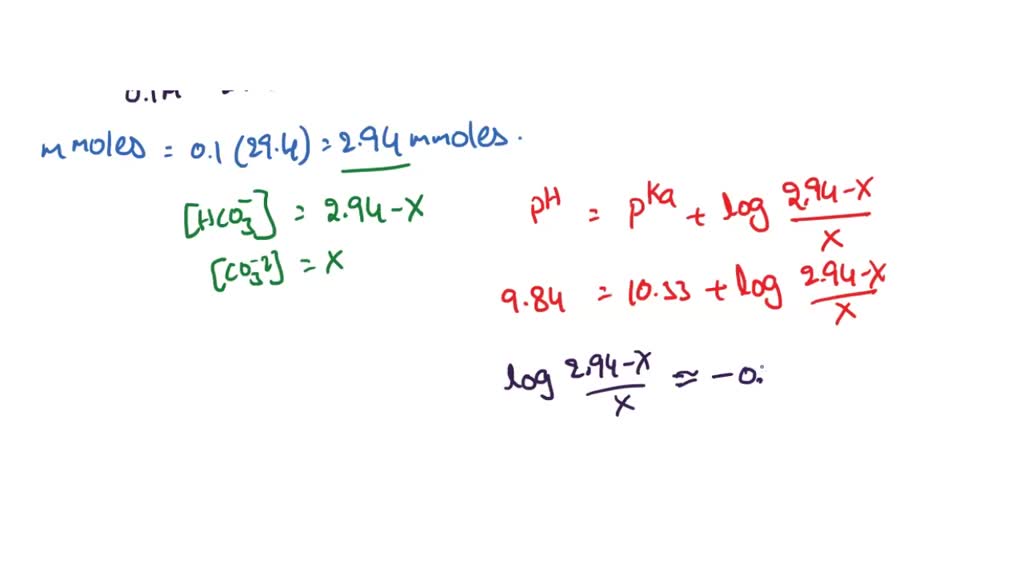
SOLVED: The pKa for the acid dissociation reaction of bicarbonate is 10.33. HCO3-(aq) + H2O(l) ⇄ CO32-(aq) + H3O+(aq) Imagine that your goal is to prepare a buffer with pH 9.84 using

Table 2 from Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar
![PDF] Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar PDF] Calculation of the buffering capacity of bicarbonate in the rumen and in vitro. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/104ba10db168516a4ba7d20867d92f2a51b891e7/3-Table1-1.png)