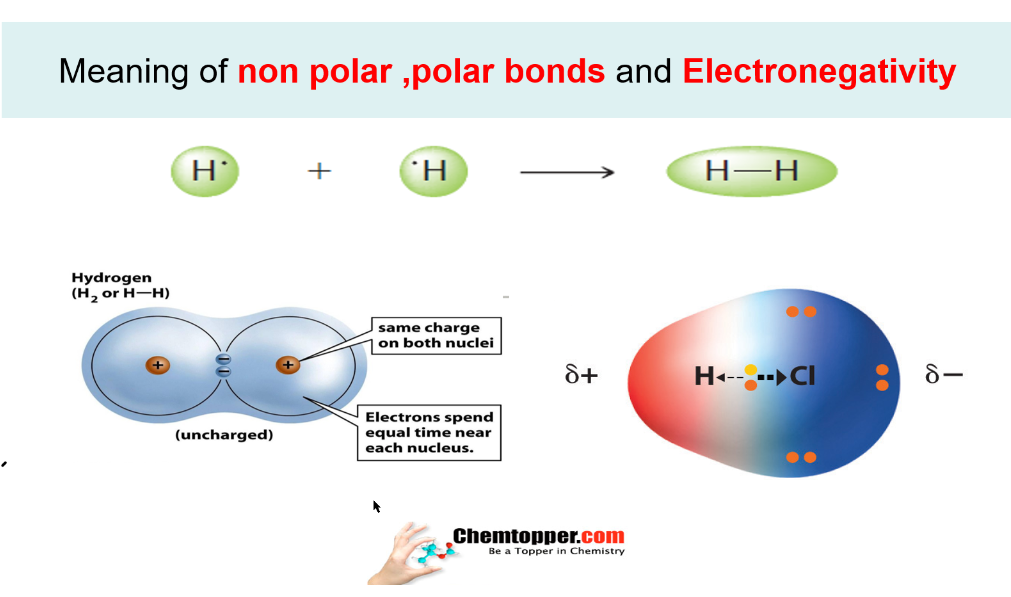
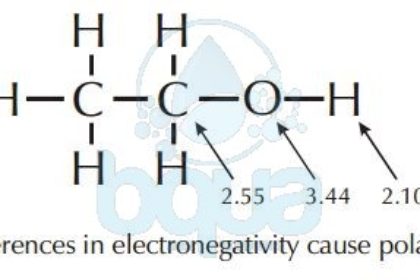
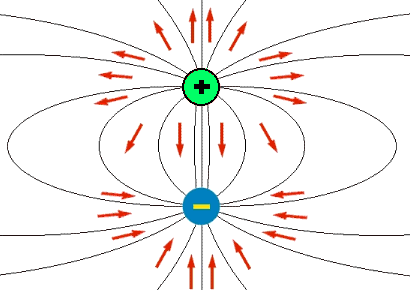
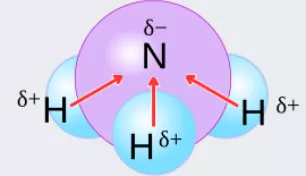
The degree of polarity of a convalent compound is measured by the dipole moment (mu(bond)) of the bond defined as: mu(bond) =Charge on one of the poles x bond length mu(bond) is

Polar Molecule Characteristics & Example | What Makes a Molecule Polar? - Video & Lesson Transcript | Study.com